Cleanroom qualification
Testo Industrial Services offers you professional support for all GMP measures in the area of cleanroom qualification. We carry out all qualification tests in accordance with Annex 1 of the EU GMP Guide, DIN EN ISO 14644 and VDI 2083.
In addition to cleanroom qualification, our cleanroom solutions for the pharmaceutical industry and medical technology also include testing and qualification of safety cabinets, isolators and other clean air systems.
We accompany you from the conception and planning of the qualification phases (DQ, IQ, OQ, PQ), through the implementation of risk management in accordance with the requirements of ICH Q9/ EU GMP guidelines, to the maintenance services and requalification measures of your cleanrooms. We support you with individual measurements or take over the complete project management.

Climate measurements
Reproducible climatic conditions are indispensable for the production of sensitive products in cleanrooms. We support you in ensuring constant temperature and humidity values in your clean room:
- Individual measurements of temperature and relative room humidity
- Carrying out climate mappings in cleanroom areas
- Definition of critical monitoring points
- Commissioning, validation and calibration of monitoring systems

Pressure measurements
The protection of cleanroom areas from contamination from less clean areas is ensured by a differential pressure concept with controlled overpressure. By measuring the pressure and the air flow, operational safety can be increased and the quality of the products ensured.
We support you with qualification measurements in your cleanrooms:
- Proof of the differential pressure cascades
- Differential pressure measurement on the filter

Flow measurements
Maintaining defined, cascaded positive pressure chambers generates air flow from areas of higher purity to areas of lower purity. By supplementing pressure measurement with measurement of air flow and flow direction, operational reliability can be increased.
We support you with qualification measurements in your cleanrooms:
- Determination of the supply air volume flow
- Calculation of the air exchange rate
- Flow visualization
- Determination of the flow velocity of TAV flows

Air purity/filter performance test
Manufacturing rooms represent the production environment and thus have a decisive influence on product quality. Air purity and filter performance must be checked regularly. The results of the measurement of airborne particles in the cleanroom in the "at rest" and "in operation" states prove whether the particle count meets the specifications for the defined cleanroom class. The filter leakage test is a metrologically complex part of cleanroom qualification and is performed in accordance with DIN EN ISO 14644-3. We support you with these tests in your cleanrooms:
- Determination of the particulate cleanliness class
- Recovery time measurements/recovery tests
- Filter leak test on HEPA filter
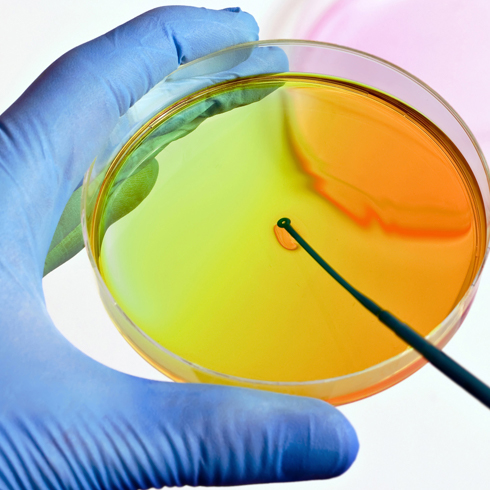
Microbiological monitoring
In addition to particle classification measurements, we also carry out microbiological sampling. We take over the complete handling of the monitoring measures from planning to the final report. The limit values for this are defined in the EU GMP guidelines Annex 1.
Our services in the field of microbiological monitoring:
- Classification measurement (microbiological)
- Testing of air (active air sampling or sedimentation)
- Testing of surfaces (direct sampling/swab test)
- Testing of personnel (in sterile areas)
- Determination of sampling points, frequencies and action limits
- Sampling
- Analysis in a GMP-certified laboratory
- Total germ count determination
- Identification of the germs (up to genus or species)
- Advice on OOS and OOT results

Medical Technology Services
Safety and compliance in medical technology
We also implement your quality assurance measures in the areas of calibration, qualification, validation and compliance in a pragmatic, cost-sensitive and effective manner in the medical technology environment.
Your advantage: Our competence
- Expertise: Consulting, measurements, project management - our industry-experienced team supports you precisely & in line with your needs
- Technology: The use of our measuring equipment with more than 4,500 references ensures reliable measurement and test results.
- Documentation: We hand over the documentation in the audit-proven layout or individually according to customer requirements to you personally on completion of the order.
GMP Training Center
Our fully equipped and GMP compliant clean room including personnel airlock offers optimal conditions for the training of our clean room staff on an area of 90 m². It is also used for the practical implementation of seminar content.
Facts about the cleanroom training centre:
- Adjustable to ISO classes 5-8 (GMP: B-D)
- Differential pressure cascade and zone concept
- Practice filter for clean room measurements
- Incl. training equipment, e.g. safety workbench, autoclave and training facility for calibrations
 Opens video in overlayThe setting of Youtube cookies is blocked until an active click on the play button is made. With this click, you give your consent for Youtube to set cookies on the terminal device you are using. You can find more information on our data protection page.
Opens video in overlayThe setting of Youtube cookies is blocked until an active click on the play button is made. With this click, you give your consent for Youtube to set cookies on the terminal device you are using. You can find more information on our data protection page.GxP-Services-Team
Professional competence, experience and high-precision equipment. The engineers and technicians of our GxP-Services Team are on duty for you throughout Europe.


Seminars
Our GMP-specific seminars provide you with in-depth practical knowledge on the topics of qualification and validation.
Success stories
We support you in your projects and are committed to your goals. Here you will find an excerpt from the references of our successful qualification projects in the cleanroom sector.

EVER Pharma Jena GmbH
Qualification of clean rooms in sterile production

Paul Hartmann AG
New cleanroom construction: project support & qualification

Pharma Stulln GmbH
Cleanroom qualification in aseptic production

At a well-known vaccine manufacturer
Qualification of pharmaceutical production facilities and cleanrooms
